Background
Several retrospective and prospective studies on TACE with LifePearlTM microspheres have been published previously. This work presents a pooled analysis of 5 registries with a total of 580 patients, that reported unresectable HCC patients treated with LifePearlTM microspheres treatment.
Objective
The aim of this multicentric, pooled analysis was to evaluate survival, efficacy, and safety in patients with
unresectable HCC treated with TACE with PEG-DEM loaded with anthracyclines.
Methods
The study included 2 prospective and 3 retrospective registries between 2015 and 2020 (see Table)
All studies enrolled patients > 18 years old with unresectable HCC that were assigned to TACE by a multidisciplinary tumour board (MDT) according to the institution's practice.
Mean age of patients was 66.9 ± 10.3 years, 72.1% were male. Distribution per BCLC stage was: 13.6% 0, 43.5% A, 41.0% B, and 1.9% C.
-
Treatment :
All studies used LifePearl TM PEG-DEM (distributed by Terumo Europe N.V., Belgium), from 100 to 400 µm. Treatment schedule, drug dosage (doxorubicin or idarubicin) and follow-up parameters was on the investigator's discretion in 4 studies or defined per protocol in 1 study.
Doxorubicin dose-escalation protocol was followed (1 study) or DEM-TACE was performed according to the clinical practice of each participating centre (4 studies)
Mean number of DEM-TACE treatments per patient was 1.89 ± 1.04. 1 TACE, 45%; 2 TACE, 32%; ≥3 TACE, 23%. Doxorubicin was used in 95.3% of cases, and idarubicin in 4.7%.
Overall survival (OS), progression-free survival (PFS), tumor response according to mRECIST (4 studies) or RECIST1.1 (1 study) criteria and analysed as best overall response), need and number of repeated TACE procedures and predictors of OS after TACE were analyzed, and safety signals were collected
| 1st Author [Ref]
| Design
| N
|
1
| De Baere [1]
| Prospective
|
97 |
2
| Veloso-Gomes [2]
| Retrospective
| 302
|
3
|
Tovar-Felice [3] | Retrospective
| 50
|
4
| Malagari [4]
|
Prospective | 26
|
5
| Verset (unpublished)
| Retrospective
| 105
|
Results
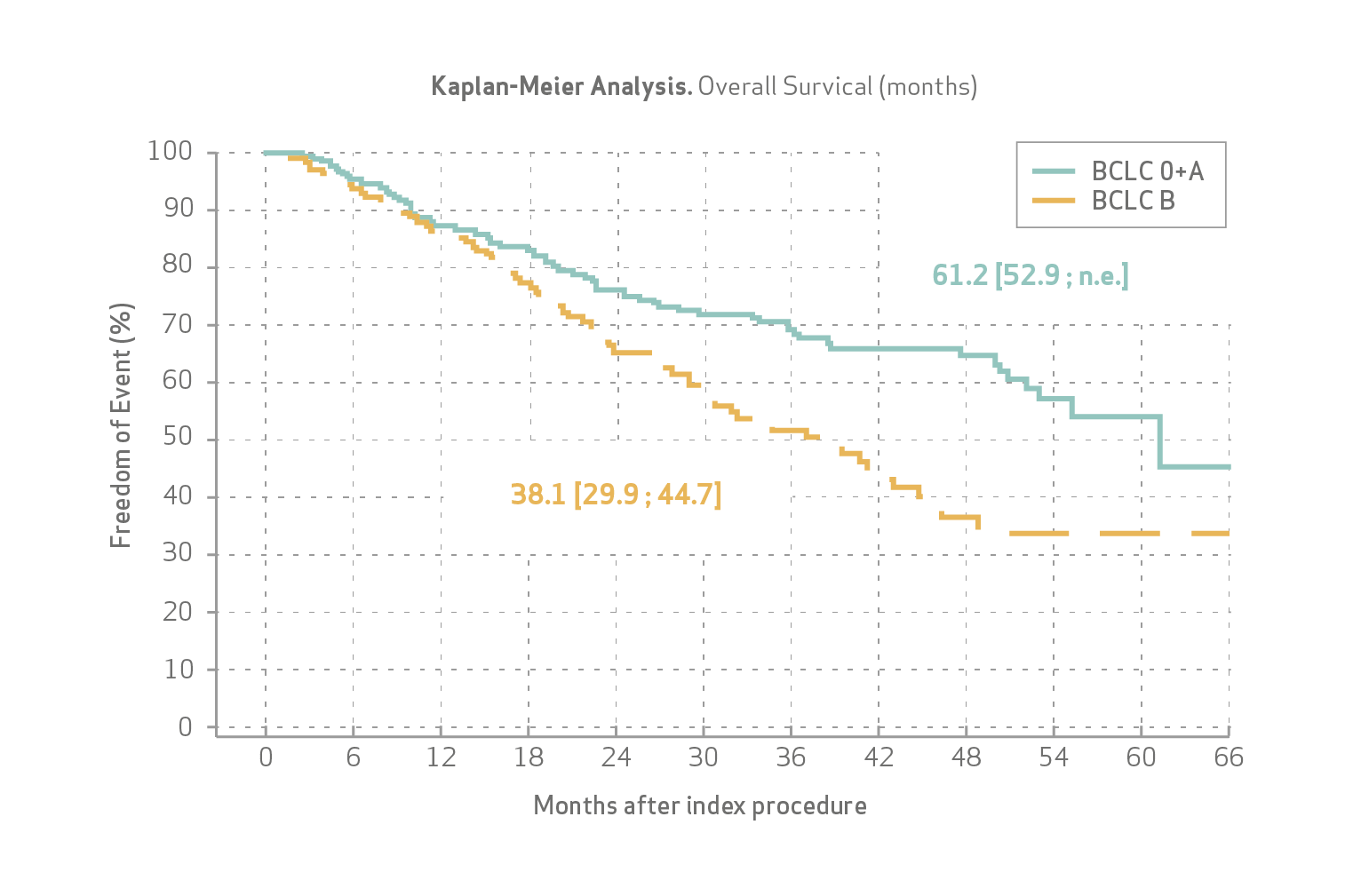
Veloso-Gomes et al pooled analysis (n=580) demonstrated:
OS was 50.8 months (61.2 for BCLC O and A, and 38.1 for BCLC B patients) (see Figure).
PFS was 15.6 months (21.6 for BCLC O and A, and 12.7 for BCLC B patients).
Median TTUP was non-estimable for the total population and patients with BCLC 0 + A, while for BCLC B stage it was 17.0 months.
Complete response (CR) was observed in 60.1% of cases, partial response (PR) in 27,1%, stable disease in 7.4% and progressive disease in 5.39%.
ALBI grade 1 and CR or PR at 3 months were the strong predictors/indicators of OS (p<0.01) in multivariable Cox regression analysis. Performance status ≥1, low baseline albumin and sum of tumour diameters were identified as negative survival predictors.
Safety : 33.8% global rate of complications was reported, mostly related with mild post-embolization syndrome with only 2.6% grade 4 AEs and 1.6% of grade 5. Image findings of hepatic damage occurred in 12.6% .
Limitations
The study was limited by heterogeneous nature of included registries and missing data points including:
Imaging time intervals and follow-up were not standardized across the studies,
Patient populations varied between the study sites
Tumour response was measured according to mRECIST in 4 studies and RECIST1.1 in one study
The survival analysis may have been biased by the inclusion of 35 patients who were treated with thermal ablation or percutaneous ethanol injection for persistent tumour.
Incompleteness of data that did not allow for time dependent analysis of survival predictors
Conclusions
Dr. Veloso et al concluded that this multicentric pooled analysis of 580 patients with HCC, treated with PEG-DEM TACE, demonstrated a median OS of 50.8 months, contributing to the knowledge of the current expected survival after TACE, for the treatment of HCC.
Key Takeaways
According to Dr. Veloso-Gomes et al publication, the data from 580 patients treated for unresectable HCC with LifePearlTM microspheres loaded with doxorubicin or idarubicin showed:
This study reports a relatively long median OS in one of the largest multicentric cohorts of HCC patients treated with TACE to date
References
- de Baere et al. J Vasc Interv Radiol 2016, 27, 1425-1431. doi: 10.1016/j.jvir.2016.05.015;
- Veloso Gomes et al. J Vasc Interv Radiol 2018, 29, 841-849. doi: 10.1016/j.jvir.2018.02.004;
- Tovar-Felice et al. Hepat Oncol. 2021, 8, HEP38. doi: 10.2217/hep-2020-0035;
- Malagari et al. J Vasc Interv Radiol. 2022 Jul;33(7):752-761. doi: 10.1016/j.jvir.2021.11.019.
