Background
The femoral artery is currently the most applied access site for PCI of complex coronary lesions, especially
when large-bore guiding catheters are required. With downsizing of transradial (TR) equipment (for example
the thin-walled radial introducer sheath Glidesheath SlenderTM), TR PCI may be increasingly applied in these
patients and might be a safer alternative compared with the transfemoral (TF) approach.
Objective
The aim of this study was to investigate whether TR PCI with Glidesheath SlenderTM is superior to TF PCI
in complex coronary lesions with respect to clinically relevant access site–related bleeding or vascular
complications.
Methods
An investigator-initiated, international prospective multicenter trial was conducted, randomizing 388
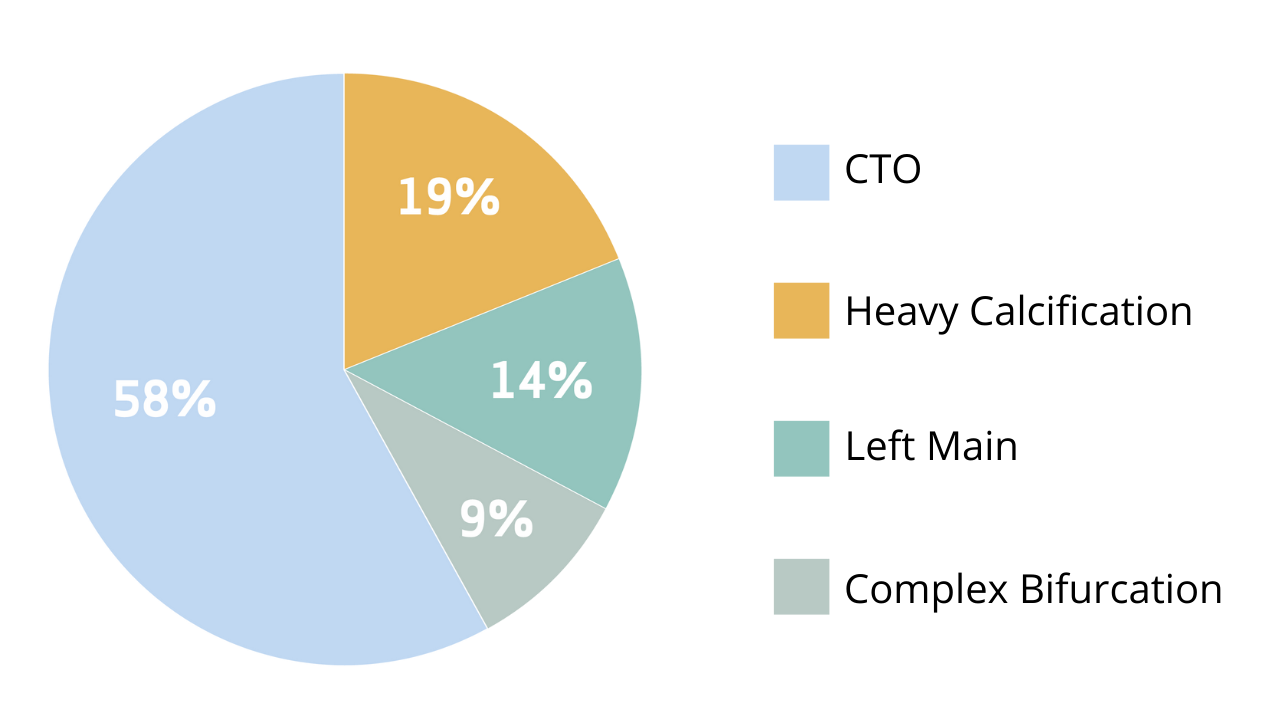
patients with planned PCI for complex coronary lesions, including chronic total occlusion, left main,
heavy calcification, or complex bifurcation, to either 7 Fr Transradial access (TRA) with Glidesheath
SlenderTM or regular 7 Fr Transfemoral access (TFA).
Procedural complexity of coronary interventions
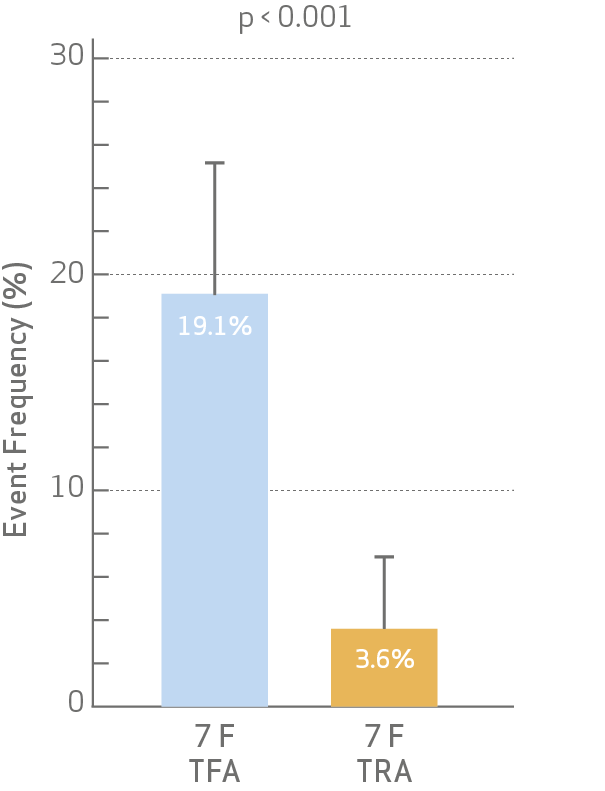
The primary endpoint was defined as clinically relevant access site-related bleeding or vascular
complications requiring intervention of the randomized access site during hospitalization.
Bleeding was classified according to the Bleeding Academic Research Consortium (BARC) criteria and
considered clinically relevant when the score was 2, 3, or 5.
As secondary endpoints TR and TF large-bore access were compared with regards to
1. Procedure-related parameters:
- Procedural success*
- Procedural time
- Fluoroscopy time and contrast volume use
- Cross-over rate
2. Bleeding or vascular complications not related to the access site
3. Major adverse cardiovascular events (MACE)**
Primary endpoint results
- The occurrence of access site-related bleeding or vascular complications at discharge was significantly lower for radial access.
BARC 2, 3 or 5 bleeding or vascular complication requiring intervention of the randomized access site
- The occurrence of BARC 2 bleeding or vascular complications were significantly lower for radial access.
Bleeding or vascular complication requiring intervention by category
Secondary endpoints results
• There was no difference between TFA and TRA with regards to procedural duration, contrast volume,
or radiation dose.
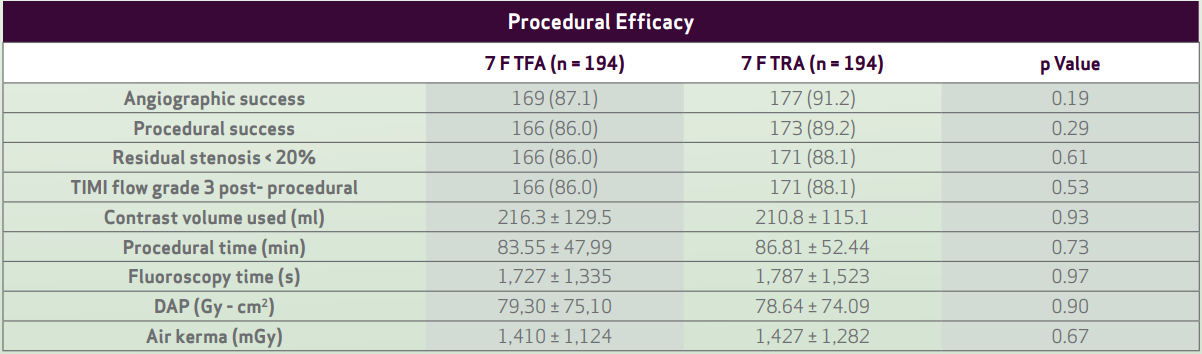
- Values are n (%) or mean ± SD
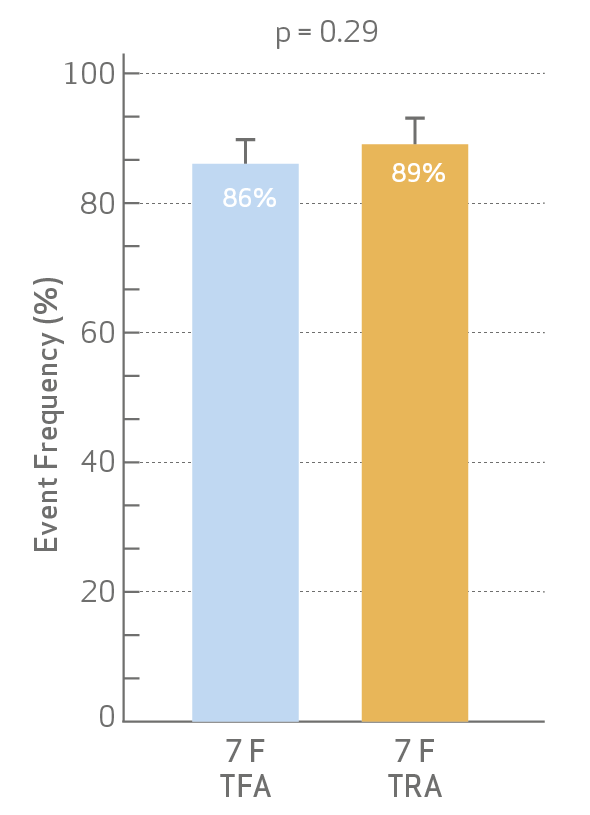
- Procedural success and cross-over
rates were not different between
TFA and TRA.
Procedural success
Crossover Rate
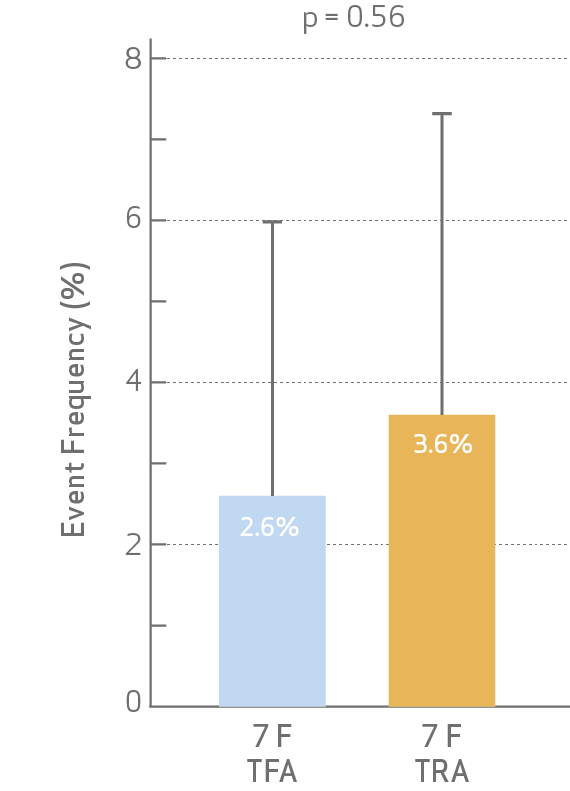
• Both combined MACE and its individual components did not reach statistical significance at discharge
and 30-day follow.

Conclusion
The COLOR Trial is the first randomized controlled trial that provides information on the use of
radial access for complex PCI lesions requiring large bore access.
The results show that radial access with 7Fr Glidesheath Slender™ is associated with lower access
site-related bleeding and vascular complications in complex PCI patients, without compromising
procedural success and other efficacy parameters. Based on these findings, radial access should be
strongly considered in all suitable patients requiring complex PCI.
However femoral access will not be completely abandoned. Further studies are required to reduce
large-bore TF access site related complications, for example with the focus on the role of ultrasound
guided puncture.
Slender technology allows performing complex procedures through radial access, allowing operators
to use regular guiding catheters, that can be used in nearly all patients.
Link to full publication: https://www.sciencedirect.com/science/article/abs/pii/S1936879821005136
References
- BARC: Bleeding Academic Research Consortium;
- CTO: Chronic Total Occlusion.
- DAP: dose-area product;
- MACE: defined as composite of death, MI and repeat revascularization of the target vessel during hospitalization and at 30 days.
- TIMI: Thrombolysis In Myocardial Infarction;
- TRA: Transradial access
- TFA: Transfemoral access
*Procedural success defined as successful PCI of the target lesion with a residual stenosis of <20%, without in-hospital MACE.
**MACE defined as composite of death, Myocardial infarction and repeat revascularization of the target vessel during hospitalization and at 30 days.
