Terumo Corporation (TSE: 4543) today announced that a new formulation to treat osteoporosis with high risk of fractures, developed by Asahi Kasei Pharma Corporation, has received approval for manufacturing and sales within Japan*. Terumo has signed a manufacturing contract with Asahi Kasei Pharma, and the upcoming product will be manufactured at Terumo Yamaguchi D&D Corporation.
The formulae, TeriboneTM 28.2㎍, is pre-filled in Terumo's PLAJEXTM syringe. Furthermore, the product is assembled as an auto-injector delivery device for a single use dosage. The unique polymer material of PLAJEX TM and Terumo's leading aseptic-filling technology was highly valued when designing this new product.
"With drug and delivery device collaboration, we are able to provide new therapeutic options to patients. We will further contribute to healthcare, by continuing to partner with pharmaceutical companies and cater to their needs", comments Tetsuya Kumei, Division President of the Alliance Division, General Hospital Company of Terumo Corporation.

TeriboneTM 28.2 ㎍ subcutaneous autoinjector
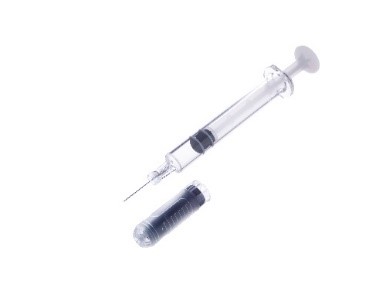
Polymer Pre-Fillable Syringe PLAJEXTM
*Asahi Kasei Pharma Press Release on September 20, 2019: "Approval to Manufacture and Sell TeriboneTM Autoinjector (interim title)"
https://www.asahi-kasei.co.jp/asahi/en/news/2019/e190920_2.html (interim URL)
About Terumo Corporation
Tokyo-based Terumo Corporation is one of the world's leading medical device manufacturers, with approximately US$6 billion in sales and operations in more than 160 nations. Founded in 1921, the company develops, manufactures and distributes world-class medical devices, including products for use in cardiothoracic surgery, interventional procedures and transfusion medicine; the company also manufactures a broad array of syringe and hypodermic needle products for hospital and physician-office use. Terumo contributes to society by providing valued products and services to the healthcare market, and by responding to the needs of healthcare providers and the people they serve. Terumo Corporation's shares are listed on the first section of the Tokyo Stock Exchange (No. 4543, Reuters symbol <4543.T>, or Bloomberg 4543: JP) and is a component of the Nikkei 225, Japan's leading stock index.
###
