Background
Despite the introduction of new chemotherapeutic agents and the development of locoregional therapies, there is still a lack of standardized evidence-based protocols for optimal Colorectal cancer with Liver metastases (CRC-LM) management
Objective
To evaluate prospectively the efficacy and safety profile of chemo embolization with drug eluting microspheres alone (DEM-TACE) vs DEM-TACE followed by bevacizumab in patients with mCRC (DEM-TACE-B).
Methods
Single center study.
Patients groups were divided per patient decision to DEM-TACE alone vs DEM-TACE + bevacizumab (DEM-TACE-B)
Treatment was performed using 2 ml of 100µm LifePearl™ microspheres loaded with irinotecan (100 mg)
For the DEM-TACE-B group, bevacizumab therapy was started 15 days after the first TACE at a concentration of 5 mg/kg and was repeated every 2 weeks for a total of eight cycles.
Tumor response was evaluated with RECIST1.1
Results
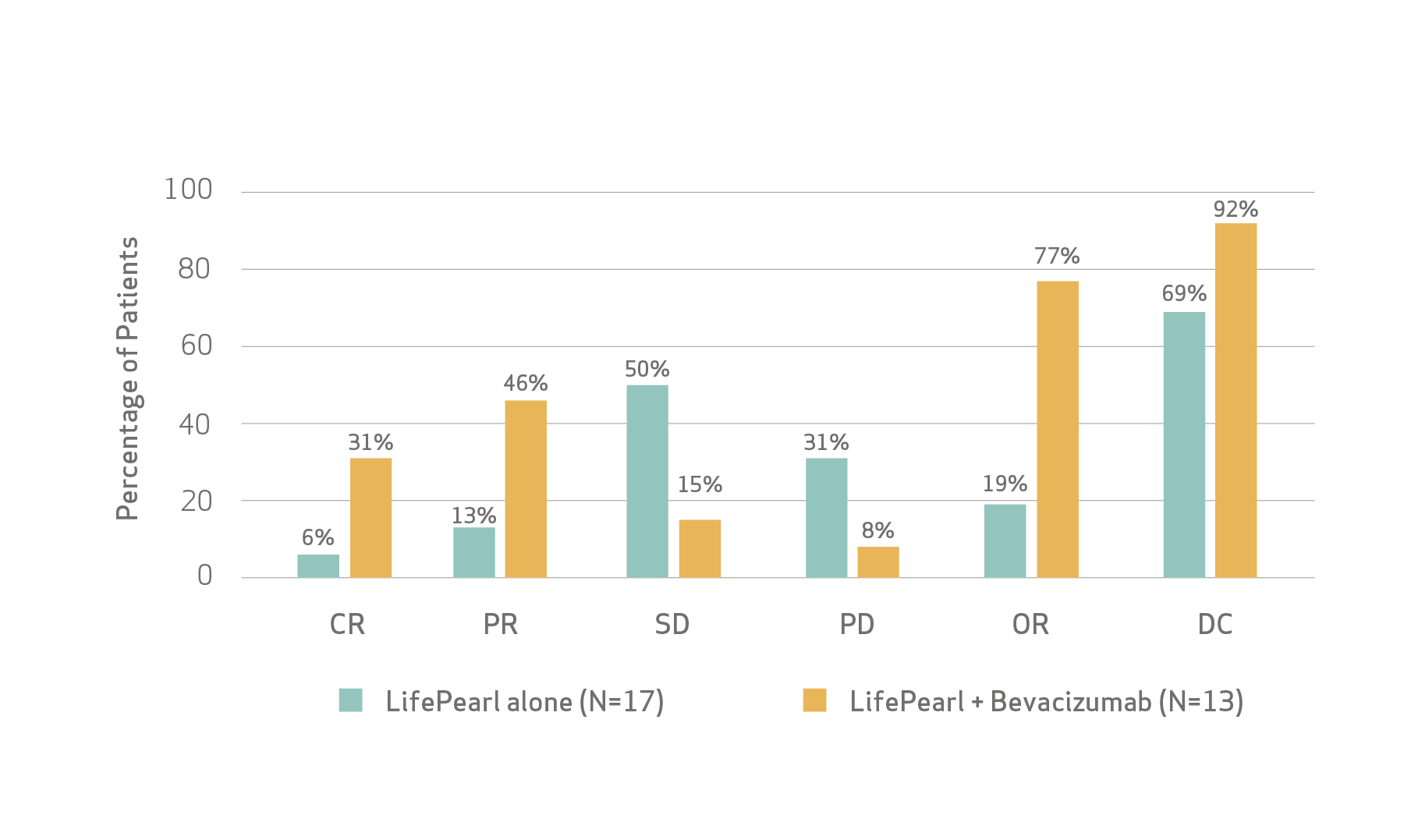
Tumor responses at 3 months
Safety profile:
Limitations
Conclusion
Fiorentini et al concluded that the results of the study suggest that the combination of TACE with intravenous bevacizumab is effective, feasible, and well-tolerated by patients with CRC-LM and may potentially have more benefits concerning tumor response and survival than TACE alone.
Key Takeaway
According to Fiorentini et al, the preliminary results of the study showed that the transarterial treatment with LifePearl microspheres loaded with irinotecan in combination with Bevacizumab is feasible and tolerable treatment with good efficacy results.
Access to the full publication: https://pubmed.ncbi.nlm.nih.gov/32111770/
European Economic Area Indications for use
LifePearl™ microspheres are indicated for embolization of blood vessels supplying primary hypervascular tumours or metastases in the liver. Note: LifePearl™ microspheres can be loaded with chemotherapeutic drugs. When used for drug loading, drug loading should be done under a physician's direction, choice and responsibility, based on type and dose of drug most beneficial to the patient. LifePearl™ microspheres are compatible with doxorubicin, epirubicin idarubicin and irinotecan. LifePearl™ microspheres can be drug loaded prior to embolization and then, as a secondary action, elute a local, controlled, and sustained dose to the targeted tumour sites after embolization. LifePearl™ microspheres are not available for sale in all countries. This information is provided only in respect to markets where this product is approved or cleared.
This literature summary is not a systemic review. It is only an example of LifePearl microspheres related literatures.
The use of the LifePearl™ devices in combination with drugs is not cleared or approved in the USA by the Food and Drug Administration. LifePearl™ microspheres are not approved in Canada. Please consult the indication of use with the IFU supplemented with the product.
Please contact your Terumo local sales representative for more information. All brand names are trademarks or registered trademarks of TERUMO CORPORATION and their respective owners. Refer to Instructions for Use for Contraindications, Warnings and Precautions.
©2021 MicroVention Europe CE0297
Manufacturer
MicroVention Europe
30 bis, rue du Vieil Abreuvoir
78100 Saint-Germain-en-Laye – France
Tel: +33(0)1 39 21 77 46
Distributor
Terumo Europe N.V.
Interleuvenlaan 40
3001 Leuven Belgium
Tel: +32 16 38 12 11
