Overview
Terumo is open to receive and to discuss study proposals to support clinical data generation of its medical devices from real world evidence, observational studies as well as from prospective studies with a focus on specific subsets of patients.
The main areas of interest for such studies are to:
- Identify the right patient population
- Evaluate safety, efficacy, feasibility, Quality of Life for these patients.
- Assess Economic aspects related to the use of our products.
Initial Proposal Application
An initial proposal application, using the protocol evaluation form, includes:
- Required supporting documents in English
- Principal investigator's curriculum vitae
- Project summary to include:
- Background
- Objective and rationale
- Primary endpoints
- Project duration
- Requested support (if any)
- Monetary (in euros)
- Materials
- Equipment
- IIS projects will be selected based on the following criteria:
- New and original study question or study idea
- Strong scientific rationale
- Adequate study design
- Focused on patient impact and clinical outcome in line with Terumo's mission - "Contributing to Society through Healthcare".
About the Investigator-Initiated Studies Program
Terumo established an internal cross-functional review process to assess the requests for support of investigator-initiated studies. A timely and complete submission will be reviewed by an internal scientific committee and Grants & Donations committee.
Terumo may support external clinical investigations with researchers who can demonstrate clear evidence of high ethical and scientific standards that govern clinical research in human subjects as stipulated by the International Council for Harmonisation (ICH) E6(R2) Guideline for Good Clinical Practice (GCP).
Investigator-Initiated Studies Program
Investigator-initiated studies have scientific and medical merit and are developed and sponsored by an independent researcher. An IIS is conducted independently without Terumo participation or involvement. The IIS researcher may request Terumo to provide any or all of the following: funding, medical device(s), laboratory test(s)/procedure(s), equipment and training.
Responsibilities of the independent researcher:
- Study design and conduct
- Protocol review and approval
- Obtaining the ethics committee and/or regulatory authority approvals, as required
- Collecting the informed consents
- Data analysis and interpretation
- Publication
- Compliance with local laws, regulations and guidelines
- Reporting to regulatory authorities
Other responsibilities include:
- Meeting specific milestones
- Submitting updates to Terumo Medical Affairs personnel
- Authoring final study report
Areas of Interest
Drug-eluting microspheres transarterial chemo embolization (DEM-TACE)
- Studies evaluating efficacy and/or safety, including combination treatments with TACE for:
- Primary liver cancer (HCC and Cholangiocarcinoma)
-
Secondary liver tumours
- Prospective clinical evidence demonstrating the benefit of using OcclusafeTM balloon catheter in liver and other organs (e.g. prostate)
Microwave Ablation - Clinical evidence demonstrating the benefit and positioning in treating lesions for:
- Lesions in liver, bone, lung, and other organs
-
Benign and/or malignant thyroid nodules
IIS Proposal Process

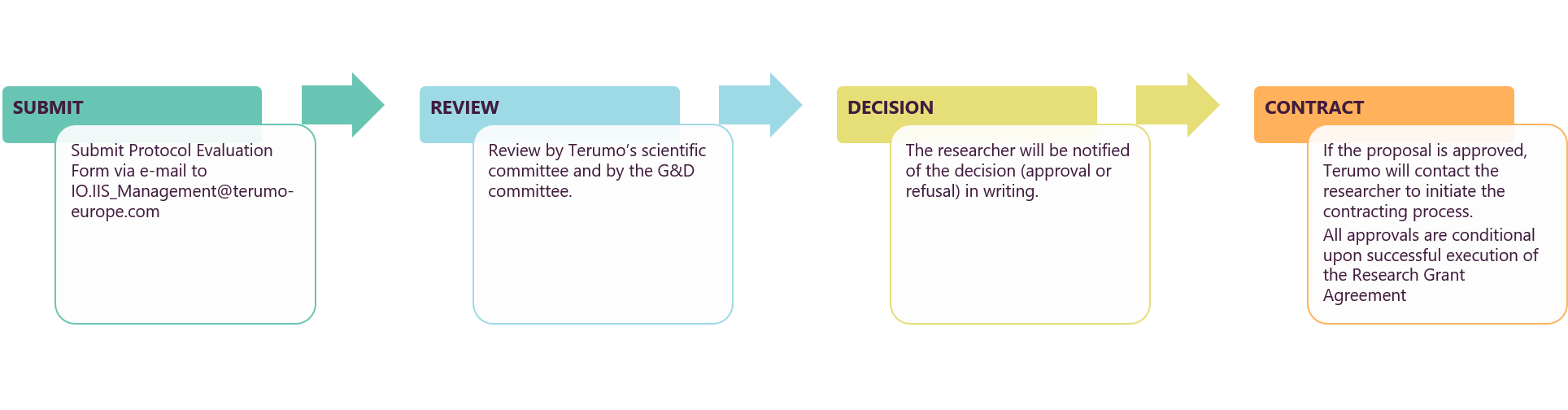
How to Submit
The independent researcher submits a fully completed and signed Protocol Evaluation Form via email to
IO.IIS_Management@terumo-europe.com
Download
