Tokyo ― Terumo Corporation (TSE: 4543 Section 1) announced CE marking for the commercial sale of the Iberis™ Renal Sympathetic Denervation System, which the company has consigned product development to Shanghai AngioCare Medical Technology Co., Ltd. (hereinafter "AngioCare") (Representative: Philip Wang), a Chinese company in renal sympathetic denervation technology for resistant hypertension treatment.
In December, 2012, Terumo formed a strategic alliance with AngioCare. Furthermore, the company has consigned product development to AngioCare and acquired the exclusive distribution rights for its renal sympathetic denervation technology on the global market.
Sales of Iberis in Europe will be commenced with its launch at the world's leading cardiovascular intervention course, "EuroPCR" to be held in France in May, 2013.
Terumo aims to expand its renal sympathetic denervation business by launching Iberis in Asian and Latin American countries where the CE marked product is allowed for commercialization.
Renal sympathetic denervation
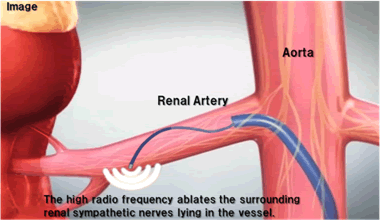
The renal sympathetic nerve plexus is a major contributor of hypertension. During renal sympathetic denervation, a catheter is introduced using standard interventional technique via the femoral artery, and is positioned in the renal artery under fluoroscopic guidance. The electrode at the distal tip of the catheter delivers high radio frequency through the wall of renal artery to ablate or disrupt the surrounding renal sympathetic nerves lying in the vessel. Renal sympathetic denervation using a catheter-based technique is highly expected to be a new, effective treatment option for the many patients with hypertension who do not respond to conventional drugs, or "resistant hypertension".
(Notice) Among the information that Terumo discloses, forecasts of financial performance on future projections contain potential risks and uncertainty since these are forecasts on projections made by Terumo based on limited information available at the moment of disclosure. Accordingly, it should be noted that actual results may differ from those forecasts on projections due to various factors. Factors affecting to actual results include, but are not limited to, changes in economic conditions surrounding Terumo, fluctuations of foreign exchange rates, and state of competition.
Any market share information in this press release is partly derived from the company's independent research.
